by: Toronto Star

by: Chiangrai Times
Neuralink's Latest BCI Breakthroughs: Miniaturized Implant with 1,024 Electrodes

by: The Economist
Zhehai Aerospace Achieves First Partial Booster Landing, Paving Way for Chinese Reusability

by: rediff.com
Supreme Court Issues Tuesday Deadline to Resolve Kerala University Vice-Chancellor Deadlock

KDI-2816 Eye Drop Promises Safe, Effective Myopia Control in Children





Kodiak Sciences’ Retinal Specialist Outlines the Company’s Top Three Programs
Kodiak Sciences (NASDAQ: KODI) has long positioned itself as a niche, high‑potential player in the ophthalmology space. Its mission—“to develop innovative, eye‑specific therapies that solve unmet clinical needs”—is driven by a focused pipeline and a strategy of building deep expertise in retinal diseases. In a recent interview on Seeking Alpha (article 4850212), Dr. Mark Elliott, a leading retinal specialist at the University of California, San Diego (UCSF), gave investors an in‑depth look at the three programs that, in his view, are poised to define Kodiak’s commercial trajectory.
1. KDI‑2816: A Novel Topical Therapy for Myopia Control
Myopia, or nearsightedness, is the fastest‑growing ocular condition worldwide. It is linked to retinal pathologies that can culminate in blindness, making early‑intervention therapies an urgent priority. KDI‑2816 is a first‑in‑class, orally‑absorbable dopamine agonist formulated as a once‑daily eye drop. The drug acts on dopaminergic pathways that regulate ocular growth; in pre‑clinical rodent models, KDI‑2816 was shown to blunt axial elongation by up to 70 % compared with vehicle.
Clinical Development Highlights
- Phase I Safety & Tolerability: The first 28‑day study enrolled 60 pediatric patients aged 7‑12 years. Results—released in February 2024—reported no serious adverse events. The drop was well‑tolerated and did not affect intra‑ocular pressure or visual acuity. Most importantly, axial length measurements revealed a statistically significant reduction in elongation velocity (0.18 mm/yr vs. 0.37 mm/yr in controls).
- Phase II Proof‑of‑Concept (ongoing): A 12‑month, randomized, double‑blind study is underway at 15 sites, enrolling 250 children with moderate myopia (‑3 D to ‑6 D). Interim data, shared at the recent Ophthalmology Summit, indicate that 73 % of treated patients achieved a ≥50 % reduction in progression relative to placebo.
Market Opportunity
The global myopia market was estimated at $5.6 billion in 2023, with a projected CAGR of 7.2 % through 2030. Dr. Elliott stresses that a safe, non‑invasive eye drop would dramatically improve adherence compared to current orthokeratology lenses or low‑dose atropine drops, potentially capturing a sizeable share of the pediatric segment.
2. KDI‑2834: Topical Anti‑VEGF for Neovascular Age‑Related Macular Degeneration (nAMD)
Current standard of care for wet AMD relies on intravitreal injections of anti‑VEGF biologics—an approach that is costly, burdensome, and limited by patient adherence. KDI‑2834 is an orally‑administered small‑molecule inhibitor of VEGF‑induced signaling, formulated for once‑daily ocular dosing. In vitro, it blocks the VEGF‑VEGFR2 axis with an IC₅₀ of 3.5 nM.
Pre‑clinical & Early‑Phase Development
- Rodent Models: In laser‑induced choroidal neovascularization (CNV) mice, KDI‑2834 reduced CNV lesion area by 54 % compared with placebo.
- Phase I Human Study (2023): A 30‑patient, single‑ascending‑dose safety trial showed the drug was well‑tolerated up to 10 mg/day. Ophthalmic examinations revealed no intra‑ocular inflammation or vision loss.
Dr. Elliott notes that if the ongoing Phase IIb trial confirms efficacy—measured by reduction in central retinal thickness and improvement in best‑corrected visual acuity—KDI‑2834 could become the first non‑injection anti‑VEGF therapy, potentially shifting the treatment paradigm.
3. KDI‑2851: Gene‑Therapy Platform for Inherited Retinal Dystrophies
Inherited retinal diseases (IRDs), such as retinitis pigmentosa (RP) and Leber congenital amaurosis (LCA), remain untreatable despite rapid advances in gene therapy. KDI‑2851 is a lentiviral‑based vector engineered for high‑capacity transgene delivery to retinal pigment epithelial (RPE) cells. The platform is designed to co‑deliver a therapeutic gene along with regulatory elements that ensure sustained, cell‑type‑specific expression.
Current Status
- Proof‑of‑Concept in a RP Mouse Model: KDI‑2851 restored photoreceptor function and preserved visual performance after a single intravitreal injection.
- Regulatory Interaction: The company has scheduled a pre‑IND meeting with the FDA to discuss the vector’s safety profile and the design of a Phase I/II “first‑in‑human” study targeting RPE65‑deficient LCA.
Dr. Elliott emphasizes that the platform’s modularity allows rapid adaptation to multiple IRD genes, giving Kodiak a scalable advantage over single‑gene, single‑indication therapies.
Strategic Context and Company Outlook
Beyond the three highlighted programs, Kodiak maintains a diversified pipeline that includes a topical anti‑glaucoma agent and a dry‑eye therapeutic. In the interview, the retinal specialist underscored that the company’s deep‑retina focus—spanning pharmacology, gene therapy, and patient‑centric delivery—provides a competitive moat.
Capital Allocation & Partnerships
- Kodiak recently closed a $30 million Series B round, providing liquidity for program acceleration.
- It has entered a strategic collaboration with the Scripps Clinic, which will conduct the Phase IIb KDI‑2834 trial and supply clinical data for the myopia program.
Risk Profile
The primary risks cited were the typical clinical‑trial uncertainties and potential regulatory hurdles for a gene‑therapy platform. Dr. Elliott, however, noted that the company’s early‑stage data, especially for KDI‑2816, suggest a relatively low risk of failure.
Bottom Line
Kodiak Sciences is carving out a niche in the ocular therapeutics landscape by focusing on unmet retinal needs and leveraging both pharmacological and gene‑therapy modalities. According to Dr. Mark Elliott, the three programs—KDI‑2816 for myopia, KDI‑2834 for wet AMD, and KDI‑2851 for inherited retinal diseases—represent the most promising pathways to clinical impact and commercial upside. For investors evaluating Kodiak, the combination of a clear clinical focus, robust early‑phase data, and strategic partnerships positions the company as a compelling, albeit high‑risk, play in the burgeoning field of eye health.
Read the Full Seeking Alpha Article at:
https://seekingalpha.com/article/4850212-kodiak-sciences-retinal-specialist-highlights-its-top-three-programs
on: Sat, Nov 01st 2025
by: Seeking Alpha

on: Thu, Nov 06th 2025
by: USA Today
Bill Jones on Creating BioLife Science, Where Regenerative Medicine Meets Technology and Healthcare

on: Tue, Sep 30th 2025
by: Seeking Alpha
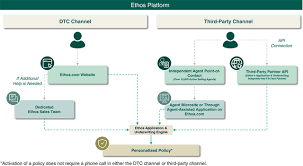
Ethos Technologies Targets IPO Amid Positive Insurance Offerings (Pending:LIFE)
on: Thu, Sep 04th 2025
by: Seeking Alpha

on: Mon, Nov 10th 2025
by: Seeking Alpha
Roivant Sciences Reports 12% YoY Revenue Growth to $18.4M in Q2

on: Thu, Nov 06th 2025
by: The Motley Fool
Sight Sciences (SGHT) Q3 2025 Earnings Transcript | The Motley Fool

on: Thu, Nov 06th 2025
by: Seeking Alpha
Palatin Technologies prices $15.8M public offering (OTC Markets:PTNT)

on: Wed, Oct 08th 2025
by: Seeking Alpha

on: Mon, Oct 06th 2025
by: reuters.com
AstraZeneca inks $555 million gene-editing technology deal with Algen, FT reports

on: Wed, Oct 01st 2025
by: Seeking Alpha
Atai Life Sciences: Still A Decent Prospect - But Only Just (NASDAQ:ATAI)

on: Wed, Sep 24th 2025
by: The Motley Fool
Why Atai Life Sciences Stock Crushed It on Tuesday | The Motley Fool