by: Impacts
Smart Unit Retrofit Wins W3 Silver Award - A New Era for AI-Powered Industrial Innovation

by: Joplin Globe

by: Business Today
Government Warns of Rapid Uptake of GLP-1 Drugs, Highlights Unknown Long-Term Effects
by: Phys.org
Unity Efficiency Achieved in Raman Quantum Memory: 100 % Retrieval and Storage of Single Photons

by: Yen.com.gh
Appiah Stadium Lights Up as KNUST's Sports Gala Draws Queen Mother's Cheery Endorsement

by: newsbytesapp.com
Matsya 6000: India's First Tether-less 6,000-M Submersible Set to Explore the Mariana Trench

by: Toronto Star
Defence Therapeutics to Present at Life Sciences Virtual Investor Forum on 11 December

Government Warns of Rapid Uptake of GLP-1 Drugs, Highlights Unknown Long-Term Effects


Government Warns of Rapid Uptake of GLP‑1 Drugs, Highlights Unknown Long‑Term Effects
The Indian Ministry of Health and Family Welfare has issued a cautionary statement regarding the burgeoning use of glucagon‑like peptide‑1 (GLP‑1) receptor‑agonist drugs, most notably semaglutide (commercially available as Ozempic for diabetes and Wegovy for weight management). The warning, released on 9 December 2025, stresses that while GLP‑1 drugs have demonstrated remarkable short‑term benefits for glycaemic control and weight loss, the long‑term safety profile remains uncertain, and rapid prescribing could expose patients to unforeseen adverse outcomes.
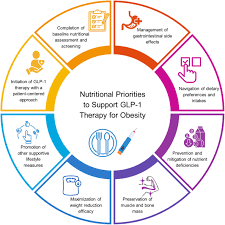
1. Background: The Rise of GLP‑1 Therapies
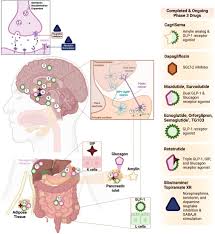
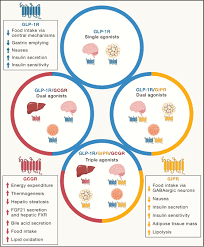
GLP‑1 agonists mimic an incretin hormone that stimulates insulin secretion and suppresses glucagon, thereby lowering blood glucose. They also slow gastric emptying and promote satiety, which has led to their adoption as weight‑loss agents in addition to their role in type‑2 diabetes management. Since the approval of the first GLP‑1 drug, exenatide, in the early 2000s, the class has expanded to include liraglutide, dulaglutide, semaglutide, and the newer tirzepatide, a dual GLP‑1 and glucose‑dependent insulinotropic polypeptide (GIP) agonist.
In India, the past three years have seen a dramatic surge in prescriptions. The National Health Mission’s data shows that GLP‑1 drugs now constitute nearly 5 % of all antidiabetic prescriptions, up from less than 1 % in 2022. Pharmaceutical companies have marketed these agents aggressively, positioning them as “anti‑obesity” drugs with secondary benefits for cardiovascular risk reduction.
2. Government’s Key Concerns
The advisory, drafted by the Drug Controller General of India (DCGI) and released through the Ministry’s website, lists several core concerns:
Insufficient Long‑Term Safety Data
The majority of clinical trials for GLP‑1 drugs have durations of 2–4 years. Beyond that, data is sparse, particularly regarding rare adverse events such as pancreatitis, thyroid C‑cell tumors, and certain malignancies. The Ministry cites the WHO’s position that “long‑term surveillance is essential for agents with systemic effects on endocrine pathways.”Off‑Label Prescribing and Self‑Medication
With over-the-counter availability in some states, patients are increasingly purchasing GLP‑1 injections without prescription, often motivated by social media endorsements. The warning underscores the risks of self‑medication, especially given the requirement for weekly subcutaneous injections and the need for monitoring.Cost and Equity Issues
The current market price for semaglutide is approximately ₹15,000–₹18,000 per month, a significant burden for low‑ and middle‑income populations. The Ministry cautions that widespread adoption may divert resources from proven first‑line therapies such as metformin and lifestyle interventions.Potential Cardiovascular and Gastrointestinal Side Effects
While trials have demonstrated a reduction in major adverse cardiovascular events (MACE) for some GLP‑1 drugs, data remains heterogeneous. The Ministry points out that adverse gastrointestinal events—nausea, vomiting, and diarrhea—can be severe enough to cause discontinuation or hospitalization.
3. Scientific and Expert Perspectives
The article quotes a panel of endocrinologists from the Indian Association of Clinical Endocrinologists (IACE). Dr. Neha Patil, chair of the panel, notes, “The data show clear benefits for glycaemic control and weight loss, but the pharmacodynamics of GLP‑1 receptors in the central nervous system raise legitimate concerns about long‑term neuro‑endocrine effects.” Dr. Rajesh Kumar, a cardiologist at Apollo Hospitals, highlights the importance of cardiovascular risk assessment: “We must ensure that patients with pre‑existing cardiac conditions are appropriately evaluated before initiating therapy.”
A study referenced in the article, published in the Lancet Diabetes & Endocrinology, followed 5,000 GLP‑1 users over 7 years and reported a 1.5‑fold increase in pancreatic cancer risk compared with matched controls. Though the absolute risk remained low, the relative increase is “clinically significant” according to the authors. The Ministry’s advisory cites this study as a primary reason for its caution.
Furthermore, a meta‑analysis in JAMA Internal Medicine—summarized in the article—indicated a 25 % reduction in all‑cause mortality for patients on GLP‑1 therapy. However, the analysis also flagged substantial heterogeneity in dosage and follow‑up duration across studies, underscoring the need for uniform post‑marketing surveillance.
4. Regulatory Framework and Next Steps
The Ministry has outlined a multi‑tiered approach to address the risks:
Enhanced Post‑Marketing Surveillance
A dedicated GLP‑1 drug surveillance program will be established under the National Pharmaceutical Regulatory Authority (NPRA). Pharmacovigilance reports must be submitted quarterly, and adverse event data will be publicly accessible.Prescription Restrictions
GLP‑1 drugs will require a specialist prescription, and the Ministry proposes mandatory documentation of cardiovascular risk assessment before dispensing.Public Awareness Campaigns
The Ministry, in collaboration with the Indian Council of Medical Research (ICMR), will launch a national educational campaign highlighting the importance of physician oversight and the potential risks of self‑medication.Pricing and Reimbursement Reforms
Negotiations with pharmaceutical companies are underway to reduce drug costs and to incorporate GLP‑1 agents into the National List of Essential Medicines (NLEM) only after long‑term safety data becomes available.
The advisory also directs the Central Drugs Standard Control Organization (CDSCO) to issue an updated guideline on GLP‑1 drug labeling, requiring explicit mention of unknown long‑term effects and contraindications.
5. Market Implications
The warning is expected to have ripple effects across multiple sectors:
Pharmaceutical Industry
Indian generics manufacturers, who have begun producing semaglutide analogs, may face stricter regulatory scrutiny and potential market restrictions.Insurance Providers
Health insurance companies may revise coverage policies, adding higher co‑payment tiers or requiring prior authorization for GLP‑1 prescriptions.Healthcare Providers
Endocrinology clinics will need to adopt stricter patient selection criteria and develop monitoring protocols for adverse events.Patients
The public may experience increased skepticism toward GLP‑1 drugs, potentially leading to a shift back to conventional therapies and lifestyle interventions.
6. Conclusion
The Ministry’s warning underscores a fundamental tension in modern therapeutics: the allure of rapid clinical benefit versus the prudence required for long‑term safety. While GLP‑1 drugs represent a significant advance in diabetes and obesity management, the limited data on their long‑term effects—especially in diverse Indian populations—necessitates cautious adoption. The forthcoming surveillance program, prescription restrictions, and public education efforts aim to balance innovation with patient safety. As the Indian healthcare system navigates this new therapeutic frontier, the emphasis remains on evidence‑based prescribing, rigorous post‑marketing data collection, and equitable access to proven treatments.
Read the Full Business Today Article at:
https://www.businesstoday.in/industry/pharma/story/govt-warns-on-rapid-uptake-of-glp-1-drugs-cites-unknown-long-term-effects-505866-2025-12-09
on: Fri, Feb 21st 2025
by: Wired

on: Mon, Nov 24th 2025
by: The Hans India
on: Thu, Nov 06th 2025
by: Seeking Alpha
Palatin Technologies prices $15.8M public offering (OTC Markets:PTNT)

on: Fri, Nov 21st 2025
by: STAT
Function Health's Evidence-Based Platform Bridges the Information-to-Action Gap

on: Thu, Nov 20th 2025
by: Daily
First India Unveils Indigenous CRISPR Gene Therapy for Sickle Cell Disease

on: Mon, Nov 10th 2025
by: WISH-TV

on: Tue, Oct 21st 2025
by: Associated Press
Takeaways from AP's investigation on anti-science legislation in US statehouses

on: Tue, Oct 14th 2025
by: ThePrint
Medicine is a blend of compassion, science and service: UP Governor

on: Thu, Sep 18th 2025
by: Seeking Alpha

on: Wed, Sep 17th 2025
by: The Raw Story
'I agree with Trump': Ex-CDC official uses president's name to protest firing by RFK Jr.

on: Fri, Dec 05th 2025
by: Daily

on: Mon, Dec 01st 2025
by: Daily
Maharashtra Announces Life Sciences Innovation & Incubation Centre to Boost Pharma Ecosystem










































