by: CNN

by: Insider

by: ThePrint
India has moved from tech follower to global leader in science and innovation: Jitendra Singh
![India has moved from tech follower to global leader in science and innovation: Jitendra Singh New Delhi, Jun 23 (PTI) Minister of State for Science and Technology Jitendra Singh on Monday said India has transformed into a global leader in science and technology over the past decade driven by a political climate that enabled scientific risk-taking and innovation. Speaking at a press conference following detailed presentations by four major [ ]](http://www.news-articles.net/images/dynamic/000/000/000/002/f9a/2.jpg)
by: SciTechDaily
Overcoming Long-Held Limitations: Korean Scientists Unveil Next-Generation Energy Storage Technology

by: MLive
Be a freshwater scientist for a day: Lake Michigan expedition mixes maritime science and history

by: Hoodline
Trump Administration Mandates 'Gold Standard Science' to Enhance Federal Research Integrity

by: Forbes
In 'The Science Of Shopping,' Kate Hardcastle Digs Into What Influences Our Purchasing Decisions

by: TechRadar

by: Hoodline

by: Variety
Academy Appoints Three New Governors-at-Large: Haifaa Al-Mansour, Effie T. Brown and Annie Chang

by: Sportschosun

by: Barchart

FDA's gene therapy turmoil unpacked

The article begins with a discussion on Leqembi, a drug developed by Eisai and Biogen for the treatment of Alzheimer's disease. Recent data presented at the Alzheimer's Association International Conference (AAIC) has shown promising results, indicating that Leqembi can slow the progression of the disease in early-stage patients. The data revealed that patients treated with Leqembi experienced a significant reduction in amyloid plaques in the brain, which are a hallmark of Alzheimer's. This reduction was associated with a slower decline in cognitive function compared to placebo. The article highlights that these findings have bolstered confidence in Leqembi's potential to become a standard treatment for early Alzheimer's, although it also notes that the drug's long-term effects and broader applicability are still under investigation.
Moving on, the article shifts focus to Gilead Sciences and its advancements in HIV prevention. Gilead has been at the forefront of developing pre-exposure prophylaxis (PrEP) medications, which are used to prevent HIV infection in high-risk individuals. The company recently announced positive results from a Phase 3 trial of a new long-acting injectable PrEP, which showed a high level of efficacy in preventing HIV transmission. This development is seen as a significant step forward in the fight against HIV, as it offers a more convenient alternative to daily oral medications. The article emphasizes the potential impact of this new PrEP option on public health, particularly in regions with high HIV prevalence.
The article also covers updates on Madrigal Pharmaceuticals and its drug Kisunla, which is being developed for the treatment of non-alcoholic steatohepatitis (NASH). NASH is a severe form of liver disease that can lead to cirrhosis and liver failure. Kisunla has shown promising results in clinical trials, with data indicating that it can significantly reduce liver fat and improve liver function in patients with NASH. The article notes that if approved, Kisunla could fill a significant gap in the treatment landscape for NASH, a condition for which there are currently no approved therapies.
In addition to these major updates, the article touches on several other developments in the biotech and pharmaceutical industries. It mentions Eli Lilly's progress with its Alzheimer's drug donanemab, which, like Leqembi, targets amyloid plaques. Recent data from a Phase 3 trial of donanemab showed that it can slow cognitive decline in early-stage Alzheimer's patients, further validating the amyloid-targeting approach to treating the disease. The article also discusses the ongoing competition between Leqembi and donanemab, noting that both drugs are vying for a share of the burgeoning market for Alzheimer's treatments.
The article further explores the regulatory landscape, highlighting recent decisions by the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). It mentions the FDA's approval of a new gene therapy for a rare genetic disorder, underscoring the agency's commitment to advancing innovative treatments. The EMA, on the other hand, is reported to be reviewing several new drugs, including a novel cancer therapy that has shown promising results in clinical trials.
The piece also delves into the financial aspects of the biotech industry, discussing recent mergers and acquisitions, as well as investment trends. It notes that several biotech companies have been acquired by larger pharmaceutical firms, a trend driven by the desire to expand product pipelines and gain access to innovative technologies. The article also highlights the increasing interest from investors in biotech startups, particularly those focused on developing treatments for rare diseases and personalized medicine.
In addition to these topics, the article provides insights into the challenges facing the biotech industry, such as the high cost of drug development and the need for more efficient clinical trial processes. It mentions ongoing efforts to streamline clinical trials and reduce costs, including the use of digital health technologies and real-world evidence. The article also discusses the importance of collaboration between academia, industry, and regulatory agencies in advancing medical research and bringing new treatments to market.
The article concludes with a look at the future of the biotech industry, emphasizing the potential for continued innovation and the development of new therapies. It highlights the role of emerging technologies, such as artificial intelligence and gene editing, in driving the next wave of medical breakthroughs. The piece also underscores the importance of addressing global health challenges, such as infectious diseases and chronic conditions, through continued investment in research and development.
Overall, the article from STAT News provides a comprehensive and insightful overview of recent developments in the biotechnology and pharmaceutical sectors. It covers a wide range of topics, from new data on Alzheimer's drugs to advancements in HIV prevention and updates on various other drugs and companies. The piece also delves into the regulatory landscape, financial aspects, and future prospects of the industry, offering a well-rounded perspective on the current state and future direction of biotech and pharmaceuticals.
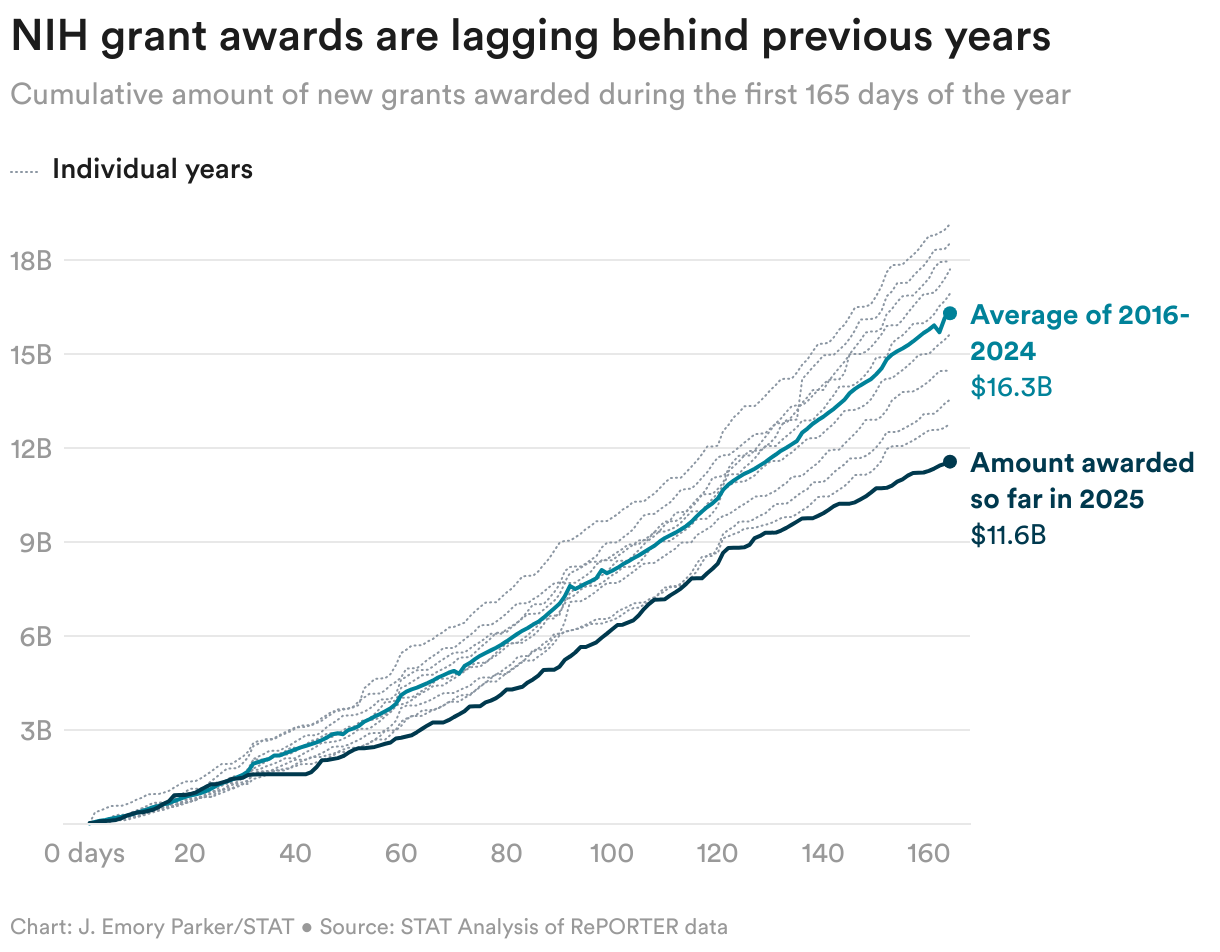
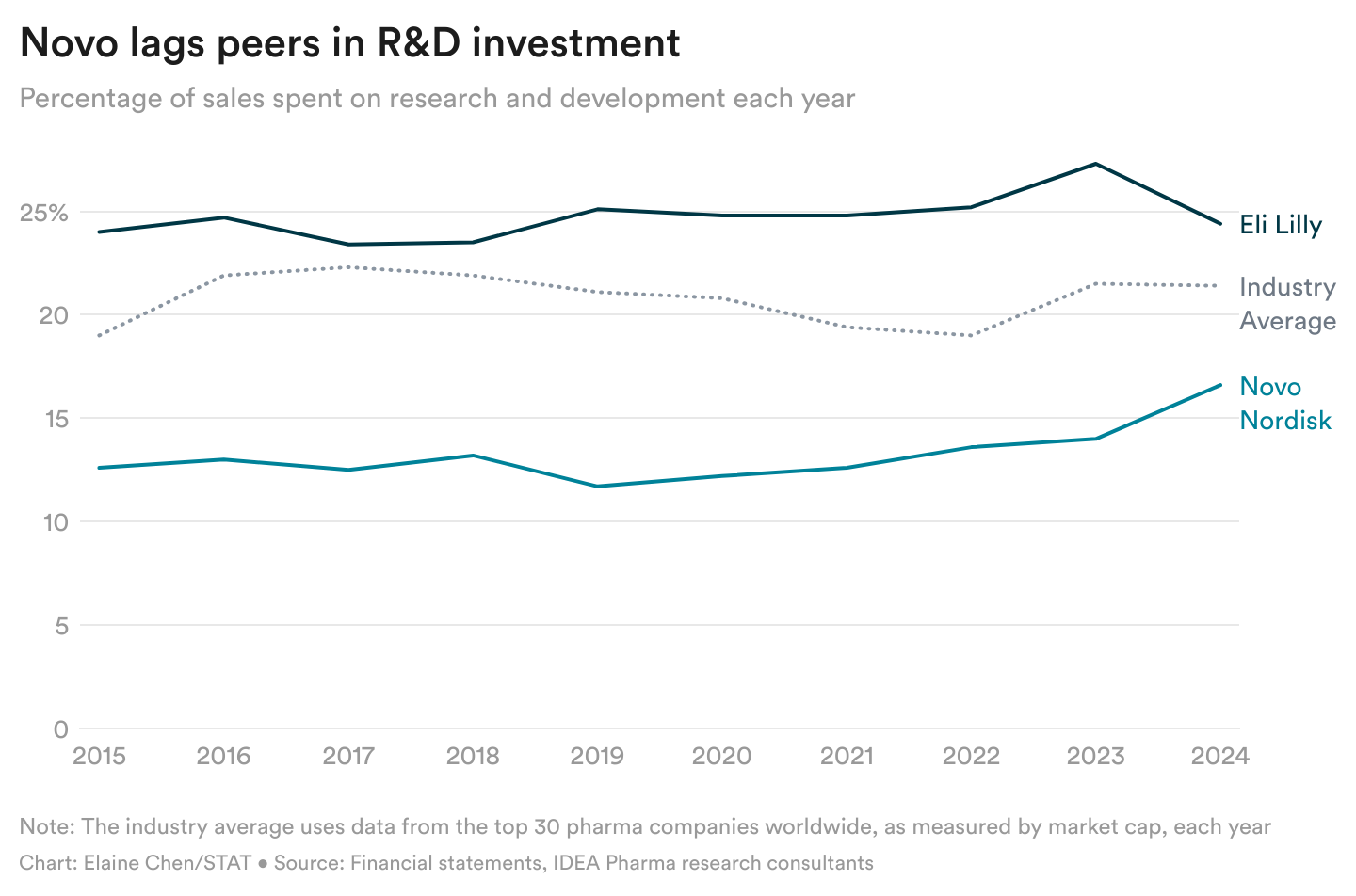
Read the Full STAT Article at:
https://www.statnews.com/2025/06/20/biotech-news-leqembi-gilead-madrigal-kisunla-eisai-eli-lilly-hiv-prep-the-readout/
on: Thu, Jun 05th 2025
by: STAT
on: Fri, Jun 06th 2025
by: STAT

on: Thu, May 29th 2025
by: STAT

on: Wed, May 21st 2025
by: STAT

on: Tue, May 13th 2025
by: STAT
Intellia cuts 27% of staff, in the latest wave of gene-editing layoffs

on: Mon, Apr 28th 2025
by: STAT

on: Tue, Apr 15th 2025
by: STAT

on: Sat, Jun 14th 2025
by: STAT

on: Wed, Jun 11th 2025
by: STAT
Merck's RSV drug approval and a drug manufacturer winds down

on: Wed, Apr 16th 2025
by: STAT

on: Thu, Feb 13th 2025
by: Forbes
How Latent Labs Plans To Create Medicines From Scratch With AI

on: Fri, Jan 10th 2025
by: FierceBiotech
Eli Lilly, Andreessen Horowitz link up on $500M biotech venture fund